Resilience Advanced Development & Manufacturing Facility
Project Details
Focus Areas & Services
Awards
Award of Merit, Jacksonville AIA
Boosting economic growth in a pro-science community
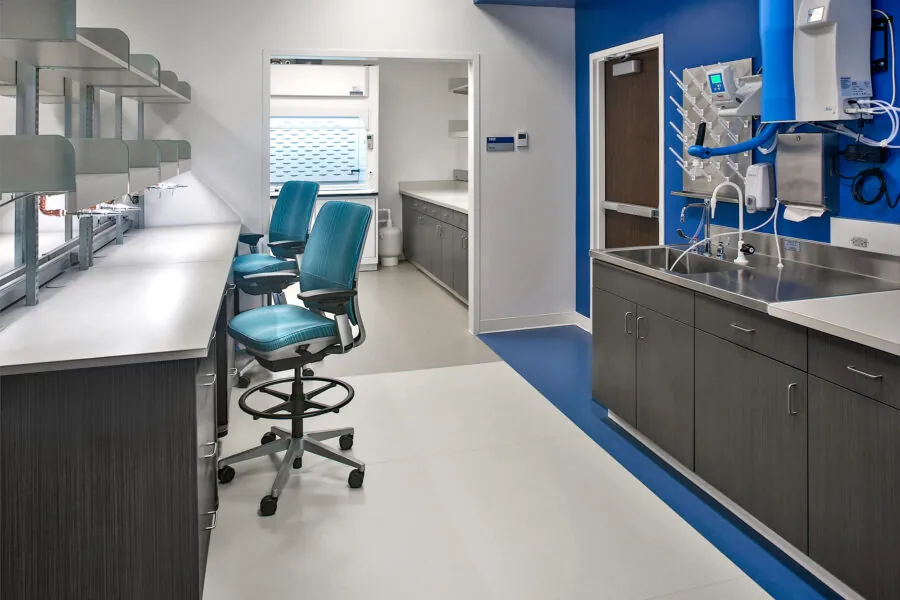
It’s not often that you get the opportunity to collaborate on a unique public-private partnership undertaking that has the potential to save lives. Building upon RS&H’s proven science facility design portfolio, the company partnered with a design-build team to design an advanced development and manufacturing facility for Resilience (formerly Nanotherapeutics and Ology Bioservices), a privately held biopharmaceutical company that sought to deliver innovative medical countermeasure products to ward off chemical, biological, radiological, and nuclear threats. Leadership from the client and design-build team worked together to submit a grant proposal that would bring financial support from the Department of Defense to fund the 165,000–square–foot advanced development and manufacturing facility that contains research and quality control laboratories, BSL-3 bio-containment laboratories and production space, pilot plant, warehouse, offices, and a conference center.
Based on the premise of keeping the warfighter in the fight, the facility required a flexible design that would allow emergency access rights to the Department of Defense should they need to take over the facility to produce potential vaccines and treatment for the military forces at a rapid, long scale. As the architect of record and laboratory programmer, RS&H developed the facility’s master plan to allow for open-ended design capabilities because it is not known what BSL-3 capability may be needed to respond to critical issues and what quantity of treatment is needed in a certain amount of time.
The site’s infrastructure, security, dual power structure, blast handling, small arms protective measures, and manufacturing functions all had to be meticulously planned and executed to meet government requirements. With so many variables in play, RS&H had to flexibly design each facility function independently, allowing for growth as needed. The building’s footprint containing six main program parts (administration, training, laboratories, process manufacturing, central utility plant, and warehouse) had to be adaptable to the extent of doubling the building’s initial square footage if necessary.

RS&H has earned a reputation for tackling complex strategic problems and delivers functional and efficient facilities. Primarily, the team focused on the health of the patient, designing state-of-the-art labs supported by a glass wash and sterilization suite, freezer bank and cryogenic storage. The production areas included ISO 7, 8, and 9 cleanrooms for BSL-3 production capabilities, including USP (Upstream) and DSP (Downstream) flexible production modules that use the latest disposable technology. Those areas are supported by personnel and material airlocks, decontamination areas and product staging and storage.
To support the critical missions of the Department of Defense, protective measures had to be put in place to validate that all building systems would be dependable, adaptable, and efficient. The building is fed by dual primary electrical feeds from separate substations and backed up by a generator and a UPS system. Multiple levels of security parameters are in place with the manufacturing areas, entries, and exits monitored by card readers and cameras. Because the local officials had never seen such a complex systems and infrastructure plan, the RS&H team had to help them learn how they work, addressing such things as a nitrogen infused fire system that conserved water, process waste flows, and how to monitor and control all the simultaneous variables at play.
Anchored by mostly startup companies in one area of town, Alachua has enjoyed substantial economic growth in the biotechnology segment and has quickly become known as a pro-science community with tax credits and incentives in place to attract businesses to locate there. With much of the community’s recent economic momentum focused on science and technology companies, high-paying jobs and an influx of a skilled labor force has been a welcomed accompaniment. Resilience’s Advanced Development and Manufacturing Facility has been a big contributor to the success story of Alachua and continues to offer viable job opportunities.
“RS&H demonstrated a deep understanding of laboratory planning principles, including both cGMP and BSL-3 level clean production areas, and BSL-3 containment laboratories. Their interior design group provided the appropriate level of finishes and workstation design that maximized utilizable space for our administrative support and training room needs. Their team provided the expertise to lead a combined effort (RS&H, Department of Defense, Resilience) to navigate between diverse regulatory requirements and practical production facility efficiencies.” -Peter H. Koury, Ph.D., MBA – President & CEO, Ology Bioservices.